Focus On Herpes Zoster

This resource is designed to offer pharmacists a concise and accurate tool to support assessing, recommending, and administering vaccines to protect against herpes zoster (often referred to as shingles). Information is included for addressing questions and concerns so that pharmacists are prepared to educate patients about herpes zoster and the risks and benefits of available vaccines.
Introduction
Herpes zoster is a vaccine-preventable disease caused by the varicella zoster virus, which also causes chickenpox.
The virus remains dormant in the dorsal root ganglion of the nervous system following the initial infection and may later manifest as herpes zoster. It is estimated that 1 in 3 people will develop herpes zoster in their lifetime, resulting in an estimated 1 million episodes in the United States annually.1
Who's at risk?
Anyone who has ever been infected with chickenpox is at risk for the development of herpes zoster. While younger adults may develop herpes zoster, it is most common in those 50 years of age and older. The risk continues to increase as adults age. People most at risk are those who are immunosuppressed.
Recognizing herpes zoster
If the virus becomes reactivated, it travels down nerve pathways from the dorsal root ganglia and erupts as skin lesions. Most commonly, the virus affects the sensory nerves within a single dorsal root ganglia, so lesions are typically observed in a dermatomal distribution (i.e., a unilateral patch of skin, usually on the face or torso, that follows a particular dermatome, an area of skin that is mainly supplied by a single spinal nerve). Symptoms of herpes zoster include a localized, painful, blistering rash. Before the rash develops, people often have pain, itching, or tingling in the area where the rash will develop. Because of the nerve involvement, patients often complain of severe neuropathic pain (shooting pain, burning, or tingling) at or near the site of the lesions. Additionally, patients may experience nausea, headache, or chills. Usually the lesions will crust over within 2-10 days, and the underlying rash will resolve in 2-4 weeks. In some instances, shingles can affect the eyes and cause loss of vision. If you suspect ocular involvement, refer the patient to a qualified ophthalmologist or physician for evaluation. On rare occasions, a patient may develop a disseminated zoster infection, which can affect a significant portion of the body (i.e., more than 1 or 2 dermatomes) or even the entire body. Disseminated zoster infections are very serious. If suspected, the patient should be advised to seek immediate medical attention.
Managing and preventing herpes zoster
There are a number of effective antiviral therapies for managing zoster reactivation, including acyclovir, valacyclovir and famciclovir. The most important factor in successfully treating a zoster infection is early detection and initiation of therapy, with the best results being achieved when treatment is started within 72 hours of symptom onset.2
Even with treatment, 10-18% of patients with herpes zoster reactivation develop postherpetic neuralgia (PHN),3 which is a debilitating and chronic pain complication of herpes zoster that can result in significant long-term morbidity, including depression, anxiety, and insomnia.4 The likelihood of developing PHN is strongly correlated with increased age at the time of herpes zoster reactivation.5
Given the potential for long-term consequences from zoster, even with prompt treatment, it is clear that the best approach to managing herpes zoster is prevention. Pharmacists can play a key role in helping patients avoid zoster reactivation and its painful sequelae by identifying at-risk patients and encouraging them to take the appropriate steps to prevent the disease, including vaccination. The remainder of this resource will assist pharmacists in understanding the available strategies for preventing zoster.
Key Counseling Points
- The first sign of herpes zoster (shingles) may not be a skin rash. Pain, itching, or tingling may precede rash development.
- Herpes zoster lesions most commonly appear on one side of the torso, but they can also appear in other locations, such as the face.
- While not common, people can get herpes zoster more than once.
- People with herpes zoster can’t spread shingles, but they can spread chickenpox.
- People can reduce their risk of herpes zoster by getting vaccinated.
Strategies for herpes zoster prevention
The HHS National Vaccine Advisory Committee (NVAC) standards for adult immunization practice encourages all health care providers to:6
- ASSESS immunization status of all patients at every visit.
- Strongly RECOMMEND vaccines that patients need.
- ADMINISTER needed vaccines or REFER patients to a provider who can immunize.
- DOCUMENT vaccines received by your patients.
Regularly performing these actions can increase immunization rates and reduce the incidence of herpes zoster as well as PHN.
Assess the Need for Zoster Vaccine
Pharmacists should regularly assess patients to determine whether they are appropriate candidates for zoster vaccine. The evaluation should consider whether zoster vaccine is indicated and whether patients have any contraindications or precautions to receipt of the vaccine.
Who should receive the zoster vaccine?
In January 2018, the Advisory Committee on Immunization Practices (ACIP) published updated recommendations for zoster vaccine:7
- Zoster vaccine recombinant (RZV) is recommended for the prevention of herpes zoster and related complications in immunocompetent adults aged ≥50 years.
- RZV is recommended for the prevention of herpes zoster and related complications in immunocompetent adults who previously received zoster vaccine live (ZVL). RZV should not be administered <2 months after receipt of ZVL.
- RZV is preferred over ZVL for the prevention of herpes zoster and related complications.
Additionally, ACIP made the following recommendations for special populations:7
- Persons with a history of herpes zoster. Herpes zoster can recur. Adults with a history of herpes zoster should receive RZV. If a patient is experiencing an episode of herpes zoster, vaccination should be delayed until the acute stage of the illness resolves and symptoms abate.
- Persons with chronic medical conditions. Adults with chronic medical conditions (e.g., chronic renal failure, diabetes mellitus, rheumatoid arthritis, chronic pulmonary disease) should receive RZV.
- Immunocompromised persons. ZVL is contraindicated in patients who are immunocompromised or are taking immunosuppressive medications. For these patients, RZV is the only currently available zoster vaccine without a contraindication. RZV or ZVL can be used safely in patients taking low-dose immunosuppressive therapy (e.g., <20 mg/day of prednisone or equivalent or using inhaled or topical steroids) and persons anticipating immunosuppression or who have recovered from an immunocompromising illness.
- Persons known to be varicella zoster virus negative. Screening for a history of varicella (either verbally or via laboratory serology) before vaccination for herpes zoster is not recommended. However, in persons known to be varicella zoster virus negative via serologic testing, ACIP guidelines for varicella vaccination should be followed. RZV has not been evaluated in persons who are varicella zoster virus seronegative and the vaccine is not indicated for the prevention of chickenpox (varicella).
What are the contraindications and precautions for zoster vaccine?
Contraindications
The following contraindications pertain to herpes zoster vaccines:8,9
- Do not administer RZV to persons with a history of severe allergic reaction (e.g., anaphylaxis) to any component of the vaccine.
- Do not administer ZVL to persons with a history of anaphylactic or anaphylactoid reaction to gelatin, neomycin, or any other component of the vaccine; persons with a history of primary or acquired immunodeficiency; persons on immunosuppressive therapy; or pregnant women or women of childbearing age.
Precautions
RZV administration should be delayed in patients with:7–9
- Current herpes zoster infection. RZV is not a treatment for herpes zoster or postherpetic neuralgia and should not be administered during an acute episode of herpes zoster.
- Pregnancy and breastfeeding. There are no available data to establish whether RZV is safe in pregnant or lactating women and there is currently no ACIP recommendation for RZV use in this population. Consider delaying vaccination with RZV in such circumstances.
ZVL administration should be delayed in patients with: 7–9
- Acute, moderate to severe illness or active untreated tuberculosis.
- Pregnancy. Pregnancy should be avoided for 3 months following vaccination with ZVL.
- Persons receiving antivirals or blood products.
ZVL and RZV are approved for use in individuals aged ≥50 years; however, ACIP currently recommends only RZV for patients aged 50-59 years. ACIP has not made any recommendation regarding the use of RZV in immunocompromised patients, but may do so in future recommendations. As previously noted, ZVL is contraindicated in immunocompromised patients.
Recommend Zoster Vaccine
If a patient is an appropriate candidate for herpes zoster vaccine, pharmacists should provide education about herpes zoster and how to prevent it through vaccination. Here are the answers to some common questions that may arise.
What causes herpes zoster (shingles)?
Both chickenpox and shingles are caused by the varicella zoster virus. After a person has had chickenpox, the virus remains latent in the nervous system, where it can be reactivated and cause shingles later in life. Approximately 30% of all people who have been infected with chickenpox will later develop shingles. The risk of getting shingles increases as patients age or become immunosuppressed.
What are the symptoms of herpes zoster (shingles)?
Shingles usually starts as a painful rash with blisters that appear in a dermatome. The rash usually clears within 2 to 4 weeks. Other symptoms of shingles can include fever, headache, chills, and upset stomach.
A subset of patients with shingles develop PHN—a form of neuropathic pain that continues after the rash resolves and can last for months or years. Very rarely, shingles can lead to pneumonia, hearing problems, blindness, scarring, brain inflammation (encephalitis), or death.
How effective is the zoster vaccine?
There are two available vaccines that have shown different levels of efficacy.
- RZV: Shingrix has been shown to reduce the occurrence of shingles by 96.6% in patients aged 50 to 59 years, 97.4% in patients aged 60 to 69 years, and 91.3% in patients aged ≥70 years. Among those aged ≥50 years, vaccine efficacy against PHN was 91.2% and 88.8% in those aged ≥70 years.8
- ZVL: Zostavax reduces the occurrence of shingles by about 70% in patients aged 50 to 59 years and by about 50% in patients aged ≥60 years; its effectiveness declines with increasing age. In a study of individuals who developed shingles despite receiving Zostavax, the duration of pain was shorter than for those who received a placebo; the severity of pain did not appear to differ among the two groups. The vaccine efficacy against PHN in patients who developed shingles was 39%.9
How safe is the zoster vaccine?
Both vaccines have favorable safety profiles.
- RZV: Common adverse reactions include mild to moderate injection site pain and possibly redness and swelling at the injection site after administration. Some people experienced fatigue, muscle pain, headache, shivering, fever, stomach pain, or nausea. Grade 3 reactions, defined as reactions severe enough to prevent regular activities, occurred in approximately 1 in 6 people. These reactions, in most cases, resolved in 2-3 days.
- ZVL: Injection site reactions (e.g., pain, erythema, swelling) were reported however fewer than 0.9% of vaccine recipients reported the symptom as grade 3. In rare instances, ZVL has been documented to cause disseminated rash and zoster in immunocompetent recipients as well as life-threatening and fatal complications in immunocompromised recipients.9
Note: If a patient experiences an adverse event following vaccine administration, it should be reported to the Vaccine Adverse Event Reporting System (VAERS) at https://vaers.hhs.gov or by calling 1-800-822-7967.
How much will zoster vaccine cost?
Depending on insurance coverage, patients’ out-of-pocket costs may vary based on the copayment structure, deductible, and other factors. Inform patients that you will check their coverage before preparing and administering the vaccine, but at the same time remind them to consider:
- Severity of shingles, including potential for PHN to cause long-term chronic neuropathic pain.
- Over the long term, the cost of receiving the vaccine may be less expensive than medical visits or medication for the treatment of shingles and PHN.
- Potential for patient to submit for direct reimbursement from insurance carrier if not covered or use of funds from health savings or flexible spending accounts.
Administer Zoster Vaccine
If the patient agrees to immunization, pharmacists who administer zoster vaccines should proceed with the immunization. Pharmacists who do not administer zoster vaccines should direct the patient to a qualified health care provider who can administer the vaccine.
Key differences between RZV and ZVL that affect their administration are listed in the chart below.
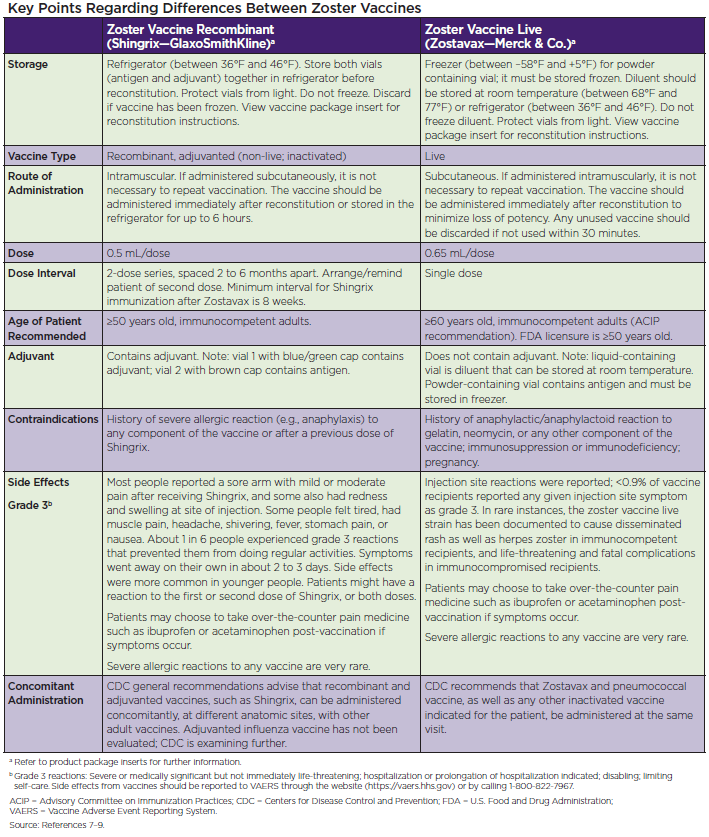
Document Zoster Vaccine
When administering the vaccine, be sure to:
- Give the patient the current Vaccine Information Statement (VIS) for the vaccine that will be administered (RZV or ZVL).
- Maintain permanent record of immunization.
a. Date the vaccine was administered.
b. Manufacturer of vaccine and lot number.
c. Anatomical site of administration and dose.
d. Date of VIS and date provided to patient.
e. Name, address, and title of the person administering the vaccine. - Provide patient with updated immunization record card.
- Report immunization to the state Immunization Information System.
- Send notification to the patient’s physician.
- Report any adverse events to VAERS and the patient’s physician.
Because RZV requires a 2-dose series, it is important for pharmacists who administer this vaccine to develop and implement a plan for recalling patients to receive the second dose in the series.
References
- Harpaz R, Ortega-Sanchez IR, Seward JF; Advisory Committee on Immunization Practices, Centers for Disease Control and Prevention. Prevention of herpes zoster: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep. 2008;57(RR-5):1–30.
- Gnann JW, Whitley RJ. Herpes zoster. N Eng J Med. 2002;347(5):340-346.
- Yawn BP, Saddier P, Wollan P, et al. A population-based study of the incidence and complication rates of herpes zoster before zoster vaccine introduction. Mayo Clin Proc. 2007;82(11):1341–1349.
- Johnson RW, Bouhassira D, Kassianos G, et al. The impact of herpes zoster and post-herpetic neuralgia on quality-of-life. BMC Med. 2010(8):37.
- Johnson RW, Rice AS. Postherpetic neuralgia. N Eng J Med. 2014;371(16):1526-1533.
- Centers for Disease Control and Prevention. Standards for Adult Immunization Practice. Available at: https://www.cdc.gov/vaccines/hcp/adults/for-practice/standards/index.ht…. Accessed February 26, 2018.
- Dooling KL, Guo A, Patel M, et al. Recommendations of the Advisory Committee on Immunization Practices for use of herpes zoster vaccines. MMWR Morb Mortal Wkly Rep. 2018;67(3):103–108.
- Shingrix package insert. Research Triangle Park, NC: GlaxoSmithKline; October 2017.
- Zostavax package insert. Whitehouse Station, NJ: Merck & Co.; September 2017.
Acknowledgments
APhA gratefully acknowledges the unrestricted financial support from Merck & Co. for the development of this resource.
The following individuals served as content and pharmacy immunization practice advisors:
- Stephan L. Foster, PharmD, FAPhA, CAPT (Ret) USPHS
Professor, University of Tennessee Health Sciences Center - Jean-Venable R. Goode, PharmD, BCPS, FAPhA, FCCP
Professor and Director, Community-Based Pharmacy Residency Program, Virginia Commonwealth University - Daniel A. Zlott, PharmD, BCOP
Vice President, Professional Education Resources
American Pharmacists Association
Online Resources
American College of Physicians, American Pharmacists Association, and The Gerontological Society of America
Aging and Immunity: The Important Role of Vaccines
http://www.navp.org/images/pdfs/FINALAgingandImmunityGuide.pdf
APhA Immunization Center
www.pharmacist.com/immunization-center
CDC Advisory Committee on Immunization Practices
http://www.cdc.gov/vaccines/acip for additional details and updates
Immunization Action Coalition
http://www.immunize.org
Zoster (Shingles) Vaccine — Guideline Summary
http://www.pharmacist.com/zoster-shingles-vaccine-guideline-summary
DISCLAIMER
Although every reasonable effort is made to present current and accurate information for public use, American Pharmacists Association (APhA) and its employees and agents do not make any warranty, guarantee, or representation as to the accuracy or sufficiency of the information contained herein, and APhA assumes no responsibility in connection therewith. The information referenced in the document is provided “as is” with no warranties of any kind. APhA disclaims all liability of any kind arising out of the use of, or misuse of, the information contained and referenced in this document. The use of information from this document is strictly voluntary and at the user’s sole risk.
