Focus On HPV

This resource is designed to offer pharmacists a concise and accurate tool to support assessing, recommending, and administering vaccines to protect against Human Papillomavirus Virus (HPV) in young adults. Information is included for addressing questions and concerns so that pharmacists are prepared to educate patients about (HPV) and the risks and benefits of available vaccines.
HPV: A Common Virus That Can Lead to Cancer
HPV—the human papillomavirus—is the most common sexually transmitted infection. An estimated 79 million Americans in their late teens and early adulthood are infected with HPV.1 Each year, nearly 14 million persons are newly infected with HPV and half of these infections occur in individuals 15 to 24 years of age. Most people (80%) will get an HPV infection in their lifetime.2 There are more than 100 types of HPV; high-risk types are associated with the development of various cancers and low-risk types can cause warts.3
HPV is spread primarily by skin-to-skin contact—usually through sexual activity. Many people who are infected with HPV do not have any signs or symptoms.3 The low-risk HPV types include 6 and 11, and these types cause 90% of genital warts. These types can also cause recurrent respiratory papillomatosis. HPV types 6 and 11 do not cause cancer. The high-risk types, however, can cause cancer and about a dozen high-risk HPV types have been identified. HPV types 16 and 18 are responsible for the majority of HPV-associated cancers, with 70% of cervical cancers caused by HPV types 16 and 18. In addition, the majority of anal, oropharyngeal, vaginal, vulvar, and penile cancers are caused by high-risk HPV types. Mouth and throat cancers are the most common HPV-related cancers in men.
The 9-Valent HPV Vaccine:
Overview
The 9-valent HPV vaccine (Gardasil 9—Merck) is currently approved by the U.S. Food and Drug Administration (FDA) for use in girls/women and boys/men 9 to 45 years of age for the prevention of a variety of HPV-associated cancers, precancerous or dysplastic lesions, and genital warts.4 On October 5, 2018, the FDA expanded the approval of the 9-valent HPV vaccine to include women and men aged 27 through 45 years. (Previously, this vaccine had been approved for use in females and males 9 to 26 years of age.) The Advisory Committee on Immunization Practices (ACIP) started discussions surrounding the expanded age group recommendations at their meeting on October 25, 2018; however, ACIP has not yet added this older age group to the official Centers for Disease Control and Prevention (CDC) adult immunization schedule. The 9-valent HPV vaccine protects against HPV types that cause cancer (i.e., HPV types 16, 18, 31, 33, 45, 52, and 58) and against the two most common types that cause genital warts (i.e., HPV types 6 and 11). Previously, there were bivalent and quadrivalent HPV vaccines on the market; however, as of the end of 2016, only the 9-valent HPV vaccine is distributed in the United States.5
The CDC estimates that 31,200 cases of cancer could be prevented with the HPV vaccine each year.2 Since the approval of HPV vaccines, infections caused by the most common HPV types associated with cancer and genital warts have dropped by 71% among teenaged girls.
Dosing Recommendations
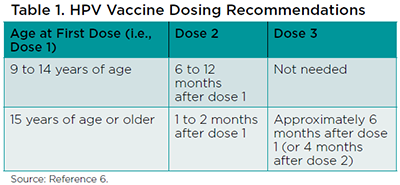
ACIP currently recommends that the HPV vaccine be given to both girls and boys starting at 9 years of age through 26 years for women and 21 years for men.6 Additionally, men 22 to 26 years of age who have sex with men should receive the HPV vaccine if not adequately vaccinated previously. The vaccine should be given routinely at age 11 or 12 years, however adults (i.e., 19 to 26 years old) who have not been vaccinated previously are still eligible to receive it. The number of doses will vary depending on when the initial dose of the HPV vaccine was given (Table 1).
The number of HPV doses needed for adult patients depends on whether they have received any doses before their 15th birthday (i.e., between 9 and 14 years of age).6 Adults who have not received any doses of the HPV vaccine before their 15th birthday should receive three doses given at 0, 1 to 2 months after the first dose, and the third dose given 6 months after the first dose. However, for those who had a dose at ages 9 through 14 years and now present to complete the series as an adult, only one additional dose is needed for a total of two doses. Adults with a history of receiving two doses of the HPV vaccine between 9 and 14 years of age, or three doses on or after their 15th birthday are considered fully vaccinated.
The reason why three doses are recommended for all patients aged 15 years and older, including adults, is that data are currently available only to assess the immunogenicity of two doses of the HPV vaccine in younger patients (i.e., 9 to 14 years of age).6 Until additional data are available in older patients for the immunogenicity of two doses, all adults who have not previously received the vaccine should be informed that three doses are needed.
ACIP also has recommendations for immunizing special populations and persons with various medical conditions.6 HPV vaccination is recommended through 26 years of age for all transgender persons and patients with medical conditions such as primary or secondary immunocompromising conditions that might reduce cell-mediated or humoral immunity (e.g., HIV infection, malignant neoplasms, autoimmune diseases). Immunocompromised persons should always receive three doses of the HPV vaccine from 9 through 26 years of age.
Recommending the HPV Vaccine to Young Adults
Pharmacists should develop a process for identifying patients who are appropriate candidates for the HPV vaccine. An assessment can be completed as part of the patient care process such as during patient intake, at the time of prescription pick-up, or through use of the immunization information system (IIS). General tips include the following:
- Identify age-appropriate patients when they are receiving other routine vaccinations, picking up oral contraceptives and emergency contraception medications, and during medication therapy management encounters, if eligible. Pharmacists should try to be proactive with education regarding HPV and offering the HPV vaccine, if appropriate in these situations.
- Individualize conversations with patients regarding the HPV vaccine to the extent possible. For example, at an outpatient pharmacy, there may be an opportunity to speak with the patient in a personal consultation area to allow for a more private discussion. Whereas, a conversation at a college health fair may be less private and result in general tips and information regarding why the vaccine is important.
- Discuss with the patient the importance of the vaccine: prevention of cancer and genital warts in both male and female individuals. Emphasize the importance of the vaccine with parents/ guardians prior to adolescents becoming sexually active in order to prevent HPV infection.
- Incorporate pharmacy staff (pharmacy technicians and student pharmacists) into the workflow to allow for additional touchpoints. Pharmacy staff can be educated on approaching eligible patients, processing vaccines through insurance, providing the vaccine administration record and Vaccine Information Statement (VIS), and drawing up the vaccine for administration. Student pharmacists can also be a significant help in administering the vaccine under the supervision of a pharmacist, if allowed by state laws and regulations.
- Technology can be used to schedule follow-up vaccinations. If possible, use existing technology from the pharmacy prescription processing database to schedule follow-up vaccine doses for patients. Send email/text message reminders to patients about their upcoming appointment, and provide them with a personal vaccination administration record. If technology is not available through the pharmacy, use free applications to schedule future vaccinations.
- Review the VIS with the patient and educate the patient on potential adverse events prior to administering the vaccine. The HPV vaccine is known to cause injection site discomfort as well as vasovagal syncope (fainting). Suggest the use of pain relievers and rotating the arm to decrease stiffness and soreness. Also, patients should be closely observed post vaccination and fall prevention strategies should be considered in case fainting occurs (e.g., having the patient seated during the vaccination).
Tips on Recommending the HPV Vaccine to Young Adults
- Be proactive to allow for prevention of HPV infection when screening patients.
- Individualize the conversation to the patient and practice setting.
- Focus on the importance of HPV vaccination to help prevent certain cancers and genital warts.
- Involve the entire pharmacy team in appropriate parts of the workflow process such as identifying patients eligible for HPV vaccination.
- Use available technology to schedule follow-up appointments and reminders to complete doses in the series.
- Provide patients with the VIS and discuss potential adverse events and mitigation strategies.
Answering Patients’ Questions About the HPV Vaccine
Older adolescents and young adult patients may be unaware that they are appropriate candidates for the HPV vaccine and may have questions. Some potential questions and suggested answers include:
Why do I need the HPV vaccine?
The HPV vaccine can help prevent infections that are associated with a variety of precancerous lesions, cancers, and genital warts.
Can HPV really cause cancer? If so, what kinds?
Yes, high-risk types of HPV can cause cervical, vaginal, and vulvar cancers in females as well as penile cancers in males. HPV can also cause anal cancer and throat cancer in both females and males.
How is HPV spread?
HPV is the most common sexually transmitted infection in the United States. The virus is spread through direct contact with an infected person. Spreading of the virus occurs most frequently with sexual intercourse; however, it can occur during sexual activity that does not involve intercourse (e.g., oral sex).
Should I still get the HPV vaccine if I’ve already had sexual contact?
Yes, the HPV vaccine can be administered to people who are already sexually active because it will protect them from HPV viruses to which they may not have yet been exposed. Ideally, people should be vaccinated before the onset of sexual activity; however, for those who have not been vaccinated, the HPV vaccine should still be given up to the recommended age to protect against other HPV types.
Should I get the HPV vaccine if I’ve never had sexual contact?
Yes, the HPV vaccine can protect individuals from HPV viruses to which they have not yet been exposed. Ideally, people should be vaccinated before the onset of sexual activity. The vaccine can be given up to the recommended age to protect against the different HPV types.
How many doses of the HPV vaccine do I need?
The number of doses depends on when the vaccine was first given and if any underlying medical conditions are present. If the vaccine was initiated between 9 and 14 years old, then two doses are needed; however, if the HPV vaccine was started on the 15th birthday or later—or if a person has an immunocompromising medical condition—then three doses are needed.
What are the most common side effects with the HPV vaccine?
The most common side effects are soreness, redness, or swelling at the site of injection. Some people may also experience a headache, muscle or joint pain, fever, or feel tired.
If I get the HPV vaccine, will I be protected against all types of HPV?
No, there are over 100 types of HPV; however, the vaccine protects against the most common types of HPV associated with cervical cancers and other types of cancers as well as the most common types of HPV that cause genital warts.
If I get the HPV vaccine, can I forgo routine cervical cancer screening?
No, routine cervical screening is still needed. The HPV vaccine does not provide protection against all types of HPV that cause cervical cancer, so even vaccinated women would still be at risk for some cancers from HPV.
I might be pregnant. Can I still get the HPV vaccine? What if I am breastfeeding?
If you are pregnant or think you may be pregnant, you should not get the HPV vaccine. Women who are breastfeeding can get the vaccine.
Will the HPV vaccine affect future fertility?
There is no current evidence that HPV vaccines cause reproductive problems in women. Both the CDC and FDA have not found any proof that HPV vaccines cause fertility issues in women such as primary ovarian insufficiency. The safety of HPV vaccines is continuously monitored to look for any new safety issues.
Who shouldn’t receive the HPV vaccine?
People with severe, life-threatening allergic reactions to a previous dose of the HPV vaccine should not get another dose. People who have a severe, life-threatening allergy to any component of the HPV vaccine should not get it. Pregnant women should not get the HPV vaccine, and people who are moderately or severely ill should wait until they are better before receiving the vaccine.
Can HPV vaccine be administered at the same time as other vaccines?
Yes, you can get other vaccines at the same time as the HPV vaccine. Administration of a different inactivated or live vaccine, either at the same visit or at any time before or after HPV vaccine, is acceptable.
Cases
Case 1.
Maria is a 19-year-old female college student who comes to the community pharmacy asking about the HPV vaccine. Maria states that she received a dose of the HPV vaccine when she was 14 years old, but received only one dose because her parents never followed up with the doctor to complete the series. She tells you she is currently sexually active and has had several partners in the past. She was wondering if she needs more doses of the HPV vaccine or if it even matters since she has already had sex with several people. What do you recommend for this patient?
- Maria should be informed that she needs an additional dose of the HPV vaccine now to complete the series. Since she received one dose of the vaccine before her 15th birthday, she only needs one more dose to complete the series (for a total of two doses).
- Although she has already been sexually active, inform Maria that she will still benefit from being vaccinated against HPV. It is possible she hasn’t been exposed to all of the types of HPV that the vaccine covers, and she would be protected from becoming infected by those types.
Case 2.
Bill is a 20-year-old male student who presents to the college health fair asking about the HPV vaccine. Bill states that he is in a committed relationship with his first and only sexual partner and was wondering whether he would benefit from the vaccine since he does not plan on being with anyone else in the future. He also tells you he has never been given the HPV vaccine in the past. What do you recommend for this patient?
- Bill should be informed that even though he has had one partner and is in a committed relationship, he is still at risk for HPV. Tell him that HPV can be acquired even if you are in a committed relationship with a single partner. It is possible that the partner may have already been exposed and never knew it since many people have HPV with no signs or symptoms.
- Educate Bill that he needs three doses of the HPV vaccine to complete the series because he has had no doses in the past. The doses should be given at 0, 1 to 2 months after the first dose, and the third dose at 6 months after the first dose.
Administer or Refer and Document HPV Vaccination
When administering the HPV vaccine (and all other vaccines), documentation of vaccine administration and communication with other members of the health care team are essential for providing collaborative care that ensures all of the patient’s immunization needs are met. Be sure to:
- Give the patient the current VIS for the vaccine that will be administered.
- Answer any questions the patient may have and discuss expectations with the patient.
- If the patient is willing to be vaccinated and you do not administer the vaccine, provide a referral (written or electronic) to a provider who will vaccinate.
- Maintain a permanent record of the immunization that includes:
- Date the vaccine was administered.
- Manufacturer of vaccine and lot number.
- Anatomical site of administration and dose.
- Date of VIS and date provided to patient.
- Name and title of the person administering the vaccine and address of the healthcare facility where the permanent record will reside.
- Provide the patient with an updated immunization record card.
- Report immunization to the state or local IIS.
- Send notification of the vaccination to the patient’s primary care provider, if known, and inform the provider that the vaccination will be submitted to the state’s IIS and the patient’s health insurance, if applicable.
- Report adverse events to the Vaccine Adverse Event Reporting System (https://vaers.hhs.gov) and the patient’s primary care provider.
- If a follow-up vaccination is needed to complete the series, inform the patient, schedule the vaccination, and/or insert the reminder need within the pharmacy’s reminder recall system.
Online Resources
CDC: Human Papillomavirus Virus (HPV)
www.cdc.gov/hpv/index.html
CDC: Immunization Schedule for Adults (19 Years of Age and Older)
www.cdc.gov/vaccines/schedules/easy-to-read/adult.html
Immunization Action Coalition: Ask the Experts—Human Papillomavirus Virus (HPV)
www.immunize.org/askexperts/experts_hpv.asp
American Cancer Society: National HPV Vaccination Roundtable
http://hpvroundtable.org
References
- Immunization Action Coalition. Ask the Experts: Human Papillomavirus. Updated October 10, 2018. Available at: www.immunize.org/askexperts/experts_hpv.asp. Accessed October 27, 2018.
- Centers for Disease Control and Prevention. 6 Reasons to Get HPV Vaccine for Your Child. May 1, 2018. Available at: www.cdc.gov/hpv/infographics/vacc-six-reasons.html. Accessed October 27, 2018.
- Centers for Disease Control and Prevention. Human Papillomavirus. Updated August 5, 2015. Available at: www.cdc.gov/vaccines/pubs/pinkbook/hpv.html. Accessed October 27, 2018.
- Gardasil 9 [package insert]. Whitehouse Station, NJ: Merck; October 2018. Available at: www.merck.com/product/usa/pi_circulars/g/gardasil_9/gardasil_9_pi.pdf. Accessed October 27, 2018.
- Centers for Disease Control and Prevention. Supplemental information and guidance for vaccination providers regarding use of 9-valent HPV. November 29, 2016. Available at: www.cdc.gov/hpv/downloads/9vhpv-guidance.pdf. Accessed October 27, 2018.
- Meites E, Kempe A, Markowitz LE. Use of a 2-dose schedule for human papillomavirus vaccination—updated recommendations of the Advisory Committee on Immunization Practices. MMWR Morb Mortal Wkly Rep. 2016;65(49):1405–8.
Acknowledgments
APhA gratefully acknowledges the financial support from Merck for the development of this resource.
The following individuals served as content and pharmacy immunization practice advisors:
Amee Mistry, PharmD
Associate Professor of Pharmacy Practice, Massachusetts College of Pharmacy and Health Sciences
Maria G. Tanzi, PharmD
Adjunct Assistant Professor, University of Illinois, College of Pharmacy
Disclaimer
APhA does not assume any liability for how pharmacists or other health care professionals use this resource. In all cases, licensed health care professionals must use clinical judgment to ensure patient safety and optimal outcomes related to HPV immunization.
